(276 W-PM) Fine-grained topography of human thalamo-cortical connectivity revealed by multi-fibre probabilistic tractography
Johannes C Klein 1, Timothy EJ Behrens 1, Matthew D Robson 2, Clare E Mackay 2,3, Matthew FS Rushworth 1,4, Stewart Shipp 5, Heidi Johansen-Berg 1
- Oxford Centre for Functional Magnetic Resonance Imaging of the Brain, University of Oxford, John Radcliffe Hospital, Oxford OX3 9DU, UK
- University of Oxford Centre for Clinical Magnetic Resonance Research, John Radcliffe Hospital, Oxford OX3 9DU, UK
- POWIC, University Department of Psychiatry, The Warneford Hospital, Oxford OX3 7JX, UK
- Department of Experimental Psychology, University of Oxford, South Parks Road, Oxford OX1 3UD, UK
- Department of Anatomy and Developmental Biology, University College London, Gower Street, London WC1E 6BT, UK
From HBM 2007 abstract site: http://www.meetingassistant3.com/ohbm2007
Parcellation of deep grey matter into functionally meaningful sub-regions has been a topic of animal research for many years. Histological studies revealed fine-grained topography of projections to different regions of cortex within individual thalamic nuclei. Due to these projections, thalamus should reflect changes in cortical organisation between species. Prefrontal cortex expanded greatly in the development of man, with mediodorsal nucleus (MD) being its main thalamic target. In pulvinar, animal research found visuotopic maps following the ventral visual stream (VVS). With the advent of multi-fibre diffusion tractography (DT), we can now aim to subdivide thalamus at an even more fine-grained level: Here, we test DT’s ability to subdivide human pulvinar and MD, mimicking macaque tracer studies by Ray and Price (2) and Shipp (1).
For a) MD, eight volunteers underwent DWI on a 1.5T Sonata (60 directions, b-value 1000 s·mm-2, 2·2·2mm3). For b) pulvinar, two volunteers underwent DWI on a 3T Trio using cardiac gating, giving 1.5·1.5·1.5mm3 voxels. Subregions of prefrontal cortex were defined in 10 separate controls using gross anatomical landmarks(Fig.1d)(3). These were then grouped following injection targets by Ray and Price (Fig.1c,d). Masks and diffusion data were spatially normalised to allow for group processing. b) DT was performed from left pulvinar to hand-drawn cortical regions in the VVS(Fig.2a). All data were processed using FMRIB’s FDT prototype.
a) Topography of prefrontal connections with human MD was similar to macaque. Putative parvicellular part of MD (Fig.1a,e:MDpv) has projections distributed to dorsal prefrontal and ventrolateral prefrontal cortex (Brodmann’s macaque areas 6,8,44,45). Orbitofrontal cortex (areas 11,12) is represented mesially in the presumed position of MD’s fibrous part (MDfi). Representation of area 46 in MD is enclosed bilaterally by both these regions, and projections to dorsal/ventrolateral prefrontal cortex encircle it(Fig.1a,e). Projections to cingulate cortex appear in caudodorsal MD (Fig1b,f:MDcd), its centre of mass being located more laterally. b) Projections to VVS line up in pulvinar along the predicted(Fig.2b), inverted rostrolateral-caudomesial gradient, with caudal cortex projecting to rostral pulvinar and vice versa.
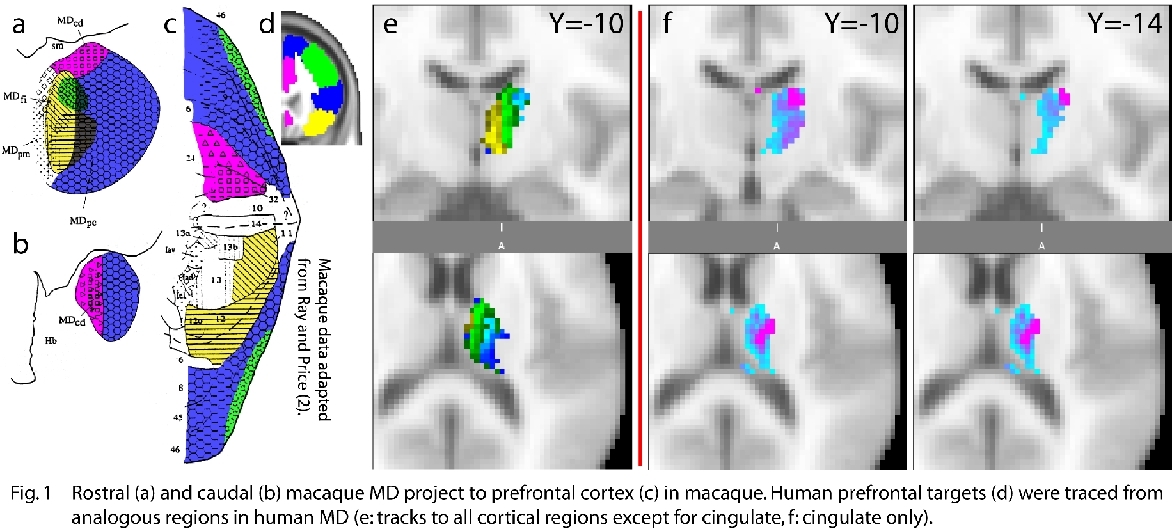
Multi-fibre DT reveals internal organisation of human thalamus closely related to macaque findings at a level unavailable with other non-invasive methods. Projections to area 46 have expanded in humans. This phylogenetically young area is not present in rodents and has expanded significantly in man relative to macaque, explaining its larger representation in human thalamus. Projections to cingulate cortex are detected in caudodorsal MD, as expected, but more laterally than macaque findings suggest. Pulvinar gradients, which proved elusive with conventional DWI, have become accessible with new high-resolution imaging methods.
In summary, multi-fibre DT establishes MD and pulvinar connectivity in concordance with monkey findings. This organisation cannot be explained by simple geometric relationships between thalamic seed locations and cortical targets. Importantly, expansion of area 46 in humans is also reflected in expansion of its representation in MD. All these findings suggest that current DT methodology is suitable for the study of sub-nuclear organisation in MD.
Shipp,S.(2004) TrendsCognSc 8
Ray,J.P.&Price,J.L.(1993) JCompNeurol 337
- Croxson,P.L.et al.(2005) JNeuroscience 25
Category = Imaging Techniques and Contrast Mechanism Subcategory = Diffusion MRI
